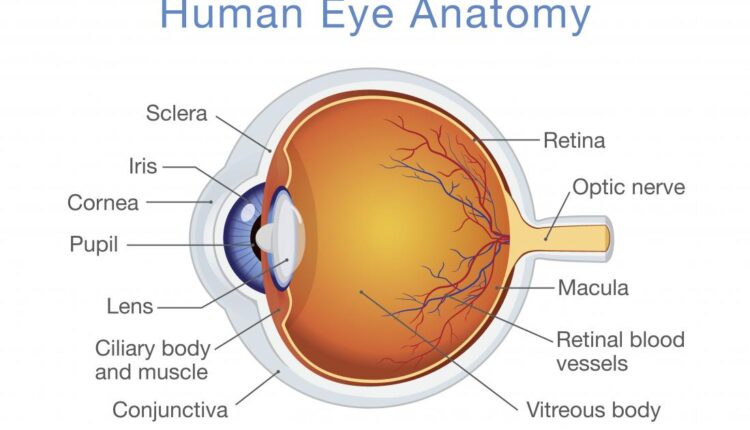
Human eye development undergoes dramatic anatomical and physiologic development throughout infancy and early childhood. Let’s have a glance over various parameters:
Dimensions of the Eye
Most of the growth of the eye takes place in the first year of life. Babies eye development begins with the change in the eye’s axial length occurs in 3 phases.
The first phase (birth to age 2 years) is a period of rapid growth: the axial length increases by approximately 4 mm in the first 6 months of life and by an additional 2 mm during the next 6 months.
During the second (age 2 to 5 years) and third (age 5 to 13 years) phases, growth slows, with axial length increasing by about 1 mm per phase. Similarly, with growth of the globe, the corneal diameter increases rapidly during the first year of life.
The average horizontal diameter of the cornea is 9.5–10.5 mm in newborns and increases to 12.0 mm in adults. The cornea also flattens in the first year such that keratometry values change markedly, from approximately 52.00 diopters (D) at birth, to 46.00 D by age 6 months, to adult measurements of 42.00–44.00 D by age 12 months.
Mild corneal clouding may be seen in healthy newborns and is common in premature infants. It resolves with premature baby eye development process the cornea gradually becomes thinner, decreasing from an average central thickness of 691 μm at 30–32 weeks’ human gestation to 564 μm at birth.
The power of the pediatric lens decreases dramatically over the first several years of life—an important consideration when intraocular lens implantation is planned for infants and young children after cataract extraction. Lens power decreases from approximately 35.00 D at birth to about 23.00 D at age 2 years. Subsequently, the change is more gradual: lens power decreases to approximately 19.00 D by age 11 years, with little or no change thereafter.
Refractive State
The refractive state of the eye changes as the eye’s axial length increases and the cornea and lens flatten. In general, eyes are hyperopic at birth and in babies eye development process it become slightly more hyperopic until approximately age 7 years, and then experience a myopic shift until reaching adult dimensions, usually by about age 16 years. Emmetropization in the developing eye refers to the combination of changes in the refractive power of the anterior segment and in axial length that drive the eye toward emmetropia. The reduction in astigmatism that occurs in many infant eyes and the decreasing hyperopia that occurs after age 6–8 years are examples of emmetropization.
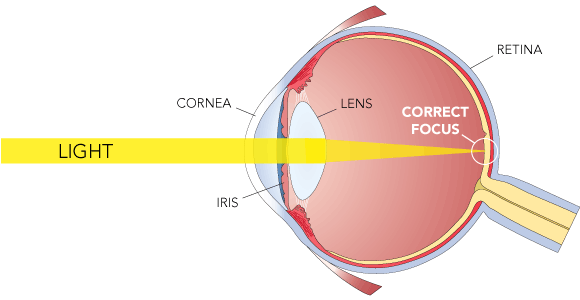
Race, ethnicity, and heredity play a role in the risk of particular types of refractive error. For example, myopia is more common among African American children compared with Hispanic children and non-Hispanic white children. Hyperopia is more common among non-Hispanic white children compared with African American and Hispanic children. Astigmatism is more common among Hispanic children and African American children than non-Hispanic white children.
Myopia is increasingly prevalent worldwide, and it is estimated that by 2050, 50% of the world population will be myopic. If myopia develops before age 10 years, there is a higher risk of eventual progression to myopia of 6.00 D or more. The etiology of increased myopia prevalence is unclear, but urbanization, increased near work, and decreased exposure to ultraviolet light are suggested influences. Low-dose (0.01%) atropine has been shown to significantly decrease myopic progression in Asian children.
Orbit and Ocular Adnexa
During baby eye development on infancy and childhood phase, orbital volume increases, and the orbital opening becomes less circular, resembling a horizontal oval. The lacrimal fossa becomes more superficial and the angle formed by the axes of the 2 orbits less divergent. The palpebral fissure measures approximately 18 mm horizontally and 8 mm vertically at birth. Growth of the palpebral fissure is greater horizontally than vertically, resulting in the eyelid opening becoming less round and acquiring its elliptical adult shape. Most of the horizontal growth occurs in the first 2 years of life.
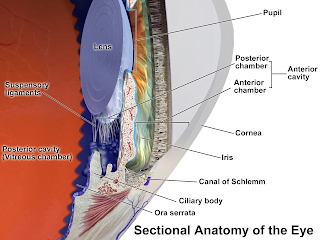
Cornea, Iris, Pupil, and Anterior Chamber
Central corneal thickness (CCT) decreases during the first 6–12 months of human eye development. It then increases from approximately 553 μm at age 1 year to about 573 μm by age 12 years and stabilizes thereafter. CCT is similar in white and Hispanic children, whereas African American children tend to have thinner corneas.
Most changes in iris color occur over the first 6–12 months of life, as pigment accumulates in the iris stroma and melanocytes. Compared with the adult pupil, the infant pupil is relatively small. A pupil diameter less than 1.8 mm or greater than 5.4 mm is suggestive of an abnormality. The pupillary light reflex is normally present after 31 weeks’ gestational age. At birth, the iris insertion is near the level of the scleral spur, but during the first year of life, the lens and ciliary body migrate posteriorly, resulting in formation of the angle recess.
Intraocular Pressure
Measurement of intraocular pressure (IOP) in infants can be difficult; results may vary depending on the method used and may not accurately represent the actual IOP. Nevertheless, normal IOP is lower in infants than in adults, and a pressure higher than 21 mm Hg should be considered abnormal. CCT influences the measurement of IOP, but this effect is not well understood in children.
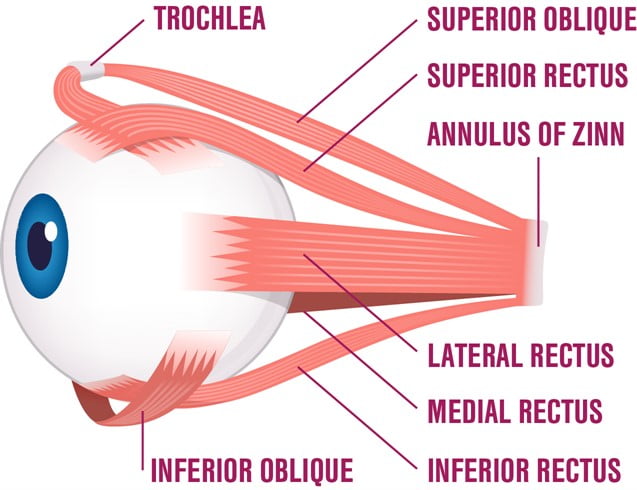
Extraocular Muscles
Baby eye muscle development stages.
The rectus muscles of infants are smaller than those of adults; muscle insertions, on average, are 2.3–3.0 mm narrower, and the tendons are thinner in infants than in adults. In newborns, the distance from the rectus muscle insertion to the limbus is roughly 2 mm less than that in adults; by age 6 months, this distance is 1 mm less; and at 20 months, it is similar to that in adults.
Extraocular muscle function continues to develop after birth. Eye movements driven by the vestibular-ocular system are present as early as 34 weeks’ gestational age. Conjugate horizontal gaze is present at birth, but vertical gaze may not be fully functional until 6 months of age. Intermittent strabismus occurs in approximately two-thirds of young infants but resolves in most by 2–3 months of age. Accommodation and fusional convergence are usually present by age 3 months.
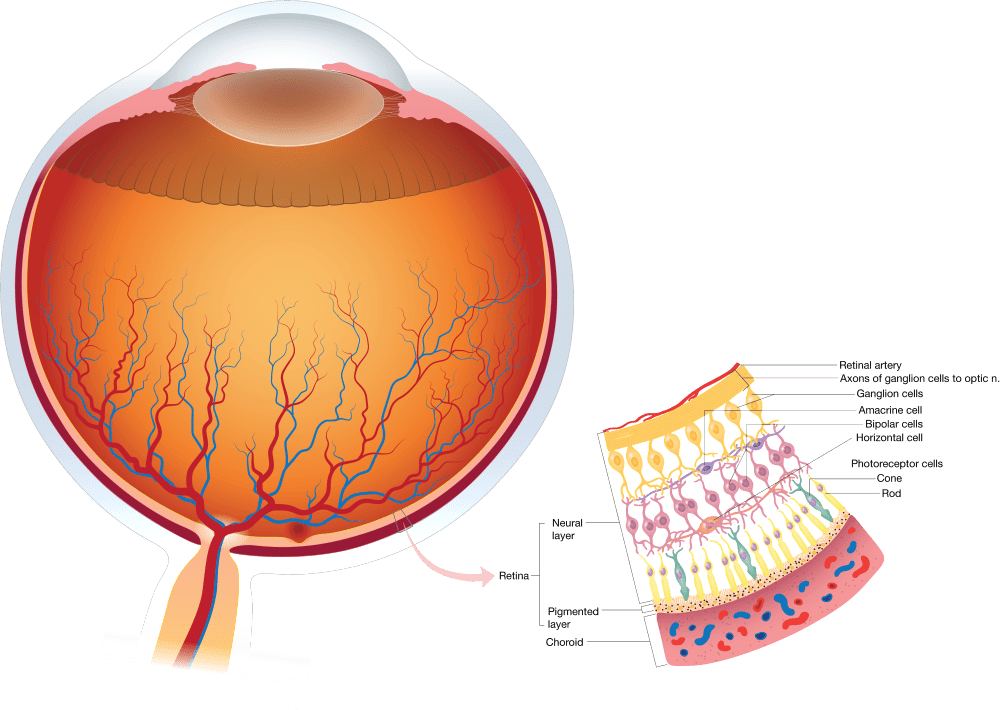
Retina
The macula is poorly developed at birth but changes rapidly during the first 4 years of life. Most significant are changes in macular pigmentation, development of the annular ring and foveal light reflex, and differentiation of cone photoreceptors. Improvement in visual acuity with age is due in part to development of the macula, specifically, differentiation of cone photoreceptors, narrowing of the rod-free zone, and an increase in foveal cone density. During retina development process retinal vascularization begins at the optic disc at 16 weeks’ gestational age and proceeds to the peripheral retina, reaching the temporal ora serrata by 40 weeks’ gestational age.
Visual Acuity and Stereoacuity
Visual acuity improvement in infancy and early childhood is attributable to neural development as well as ocular structural changes. Two major methods are used to quantitate visual acuity in preverbal infants and toddlers: preferential looking (PL) and visual evoked potential (VEP).
Studies show that visual acuity improves from approximately 20/400 in newborns to 20/20 by age 6–7 months on human eye development process.
However, PL studies estimate the vision of newborn is to be 20/600, improving to 20/120 by age 3 months of vision development in newborns and to 20/60 by 6 months. Further, PL testing shows that visual acuity of 20/20 of baby vision is not reached until age 3–5 years. The discrepancy between measurements obtained by these 2 methods may be related to the higher cortical processing required for PL compared with VEP. Stereoscopic acuity reaches 60 seconds of arc by about age 5–6 months.
(Source: AAO)